Abstract:
Background: Myeloid neoplasms with germline predisposition comprise a heterogeneous group of disorders caused by deleterious inherited variants in genes conferring susceptibility to hematologic malignancies¹. Germline variants in DDX41, located on chromosome 5 and inherited in an autosomal-dominant pattern, account for ~3% of myeloid malignancies and typically present in the sixth decade (range 44–70 years) as apparently sporadic myelodysplastic syndrome or acute myeloid leukemia. Clinical features include cytopenia, macrocytosis, and bone-marrow dysplasia with or without blast excess, usually with normal karyotype and immunophenotype²?³. International guidelines recommend identification of pathogenic/likely pathogenic variants, genetic counselling, and family screening???. Although hundreds of germline DDX41 variants have been described, the clinical significance of many missense variants remains uncertain???.
Aims: To investigate the clinical significance of the DDX41 variant c.1079C>T/p.(Thr360Ile) (NM_016222.4) through familial segregation analysis in a carrier family.
Methods/Results: A 32-year-old woman with IgA deficiency and persistent eosinophilia (>1500/µL) associated with monocytosis, without cytopenias or blasts, underwent bone-marrow evaluation showing no dysplasia or blast excess. Secondary and clonal causes of eosinophilia were excluded, including PDGFRA, PDGFRB, FGFR1, JAK2, and BCR-ABL rearrangements; karyotype was 46,XX.
Targeted NGS identified the DDX41 variant c.1079C>T/p.(Thr360Ile) with a variant allele frequency of 51%, and germline origin was confirmed in peripheral-blood CD3? lymphocytes. The variant is reported in GnomAD with a population frequency of 0.0032% (rs1284223764) and in ClinVar (ID1337724), where it is classified as a variant of uncertain significance. In silico predictors are inconclusive, although the substitution affects a moderately conserved residue within the DEAD-box domain. According to ACMG/AMP criteria?, it meets PM2_supporting and PM1_moderate evidence and was therefore classified as class 3 (VUS).
The variant had previously been described as a somatic mutation in one patient with acute myeloid leukemia?, prompting a segregation study. Familial testing identified the variant in the patient’s mother and maternal aunt (66 and 70 years), neither of whom shows cytopenias, dysplasia, or blast excess; the variant was absent in her sister. Germline pathogenic/likely pathogenic DDX41 variants are established drivers of inherited susceptibility to myeloid neoplasms. Although penetrance is incomplete, clinically overt disease—characterized by cytopenias, dysplasia, or increased blasts—typically manifests between 44 and 70 years. The absence of hematologic abnormalities in relatives at the upper end of this age range suggests limited clinical impact or reduced penetrance of the p.Thr360Ile substitution. Accordingly, the variant remains classified as a VUS, and further functional and segregation studies are required to clarify its pathogenic role.
Conclusion: In this family, the germline DDX41 p.Thr360Ile variant lacks evidence of a demonstrated pathogenic role in inherited predisposition to myeloid neoplasms?. This case underscores the diagnostic and counselling challenges posed by rare germline missense variants in DDX41 and highlights the need for functional studies, epidemiologic validation, and extended familial co-segregation analyses to enable definitive reclassification.
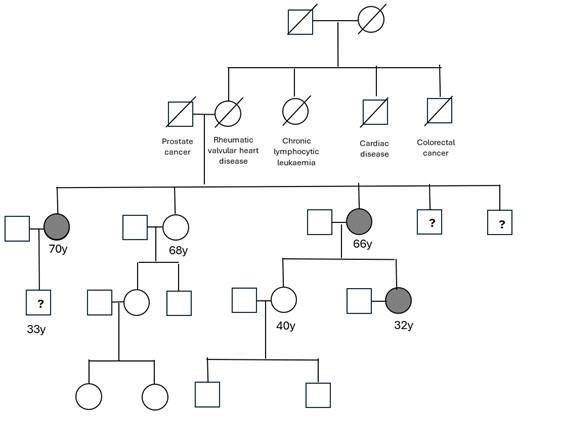
Figure. 32-year-old female; ages and causes of death are provided where applicable. Question marks denote living, untested individuals.
Biography:
Dr. Ada Esteban-Figuerola is a consultant hematologist at Hospital Universitario San Pedro (La Rioja, Spain), specializing in diagnostic hematology and myeloid neoplasms. She completed her residency in Hematology at Hospital Joan XXIII (Tarragona) and holds advanced postgraduate degrees in hematopoietic transplantation, myelodysplastic syndromes, and acute leukemias. Dr. Esteban-Figuerola is an active member of PETHEMA, GESMD, and GECH (SEHH), with a strong focus on integrated morphologic and molecular diagnosis. She has authored numerous peer-reviewed publications and congress presentations and is actively involved in teaching and multidisciplinary molecular pathology programs.
Copyright 2024 Mathews International LLC All Rights Reserved